Summary
I think Roivant shares could garner over 100% upside with minimal downside at current levels, an opportunity presented because biotech has become an investment hellscape where market participants are scared of their own shadows. It’s exactly the type of environment in which a value investor can thrive, and I believe ROIV is an extreme case of dislocation from fundamental reality.
Background
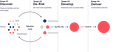
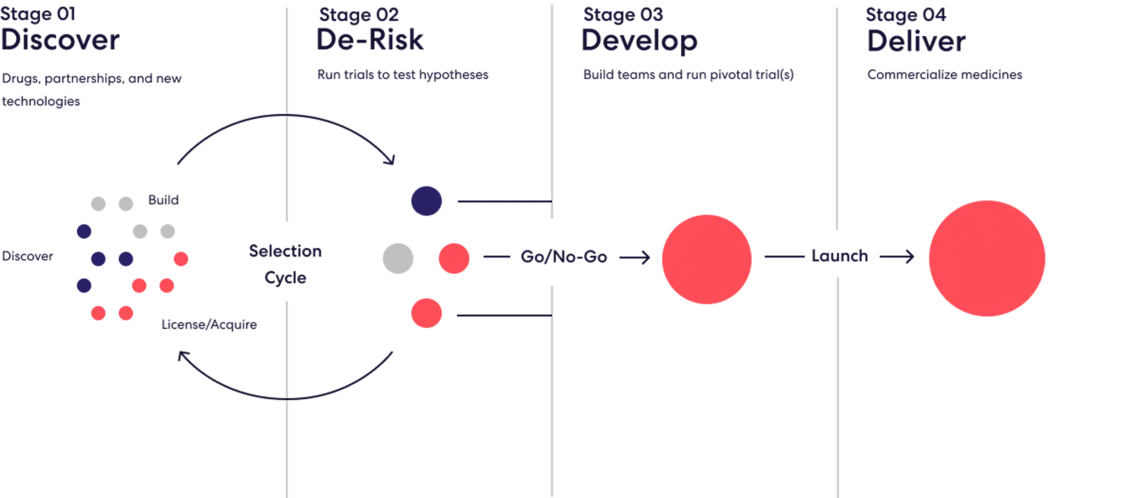
Roivant ROIV 0.00%↑ was founded in 2014 by Vivek Ramaswamy (not involved anymore) with an idea to incubate a number of subsidiaries (“Vants”) in different therapeutic modalities while applying a consistent, mercenary capital allocation policy across all of them. The process, which you can see in action as we walk through the various Vants, is depicted below:
Vivek stepped down in 2021, the same year ROIV went public via a SPAC transaction with Patient Square’s SPAC. Due to Vivek’s increasing political aspirations he stepped off the board in early 2023.
Since that time, the company has been led by Matt Gline who joined in 2016 as CFO and who has a somewhat non-traditional biotech CEO background, having been an investment banker at Goldman and Barclays/Lehman, with a focus on fixed income structuring and risk management. Relative to other CEO’s, he seems thoughtful and pragmatic, notable in a sector whose CEOs appear to have checked their pragmatism at the door chasing lottery ticket wins funded by a belief in unlimited equity funding. He transacted Datavant in 2021 through the merger with CIOX, has been instrumental in in-licensing the drug that is now IMVT-1402 in 2022, and most recently orchestrated the TL1a deal with Pfizer and subsequent exit to Roche for a 110x return.
The Major Subsidiaries (Vants)
Immunovant
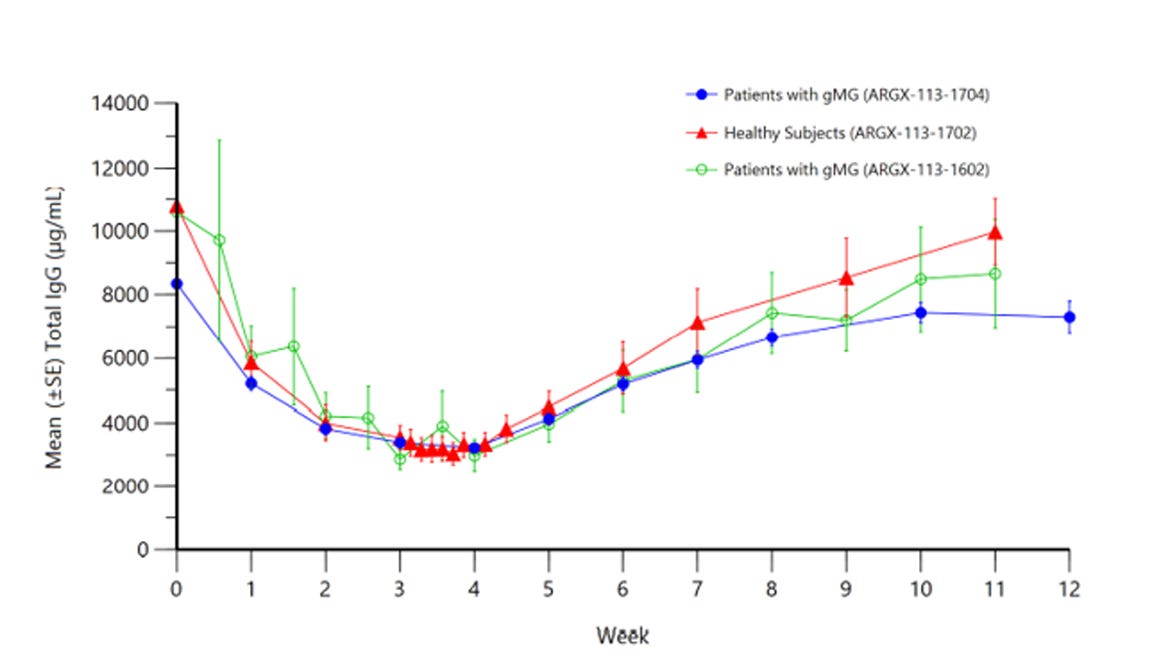
Immunovant (IMVT) stands out as the key asset in the ROIV portfolio, essential for its strategic flexibility. This significance stems from IMVT's development of drugs targeting FcRn, a receptor implicated in autoimmune diseases. These drugs aim to reduce IgG antibodies, which, in excess, can harm healthy tissue. The market already has two successful FcRn inhibitors: Argenx's VYVGART and UCB's RYSTIGGO. Notably, IMVT's leading drug candidate, IMVT-1402, has shown promising results in lowering IgG levels without the adverse effects seen in its predecessor, IMVT-1401.
ROIV's majority ownership in IMVT is a critical factor, influencing both development decisions and financial implications. The company's share price has seen fluctuations, particularly following safety concerns around IMVT-1401. However, the recent progress with IMVT-1402, exhibiting significant IgG suppression, renews optimism. This potential for efficacy, comparable or even superior to existing treatments, positions IMVT as a strong player in the autoimmune treatment landscape.
The investment opportunity in IMVT lies in its ability to address various autoimmune conditions, leveraging its FcRn inhibition mechanism. This approach could challenge the current IVIG therapies, which have a substantial global market. Furthermore, with ROIV's significant stake, a decision to sell IMVT could result in considerable financial gains, given the promising market outlook and the success of existing FcRn inhibitors.
Priovant
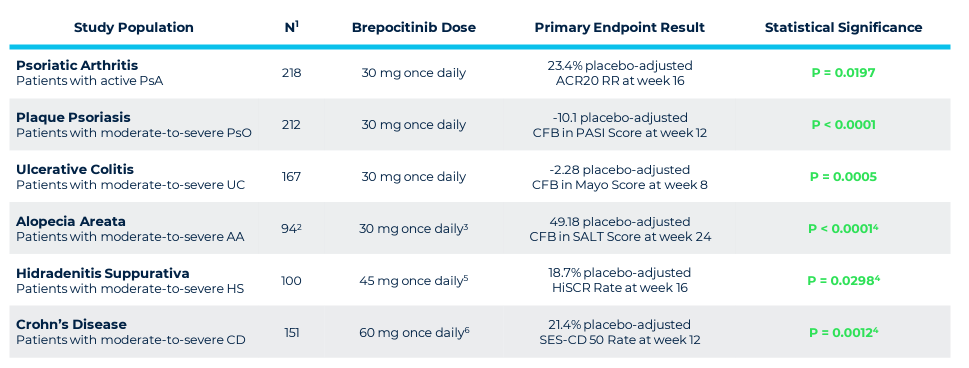
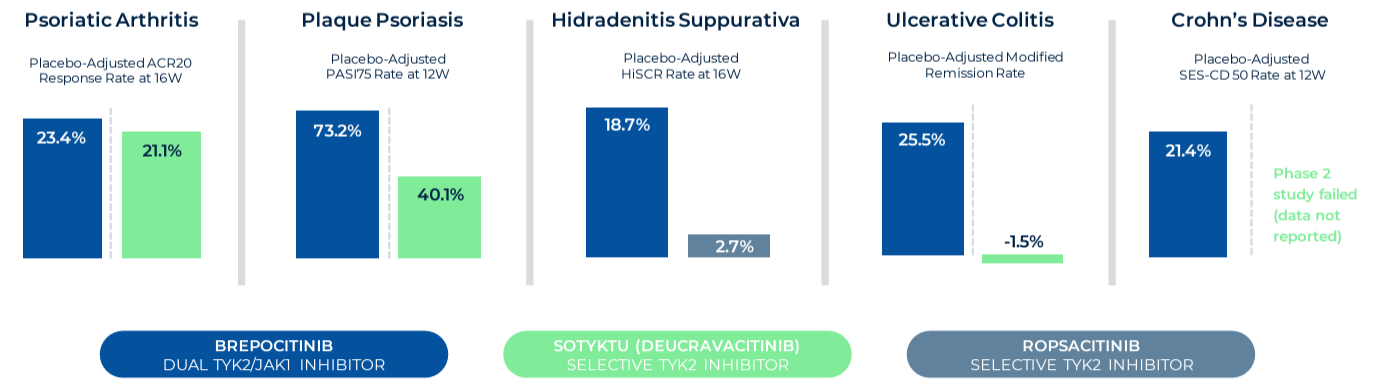
Priovant represents a venture with a high-risk, high-reward profile, in collaboration with Pfizer. Its primary focus is Brepocitinib (Brepo), a drug targeting inflammatory diseases through TYK2 and JAK1 inhibition. Despite the failure in a lupus trial, Brepo's efficacy in other conditions and its potential market advantage make it a notable asset. However, challenges like a late market entry and regulatory warnings associated with JAK1 inhibitors affect its valuation.
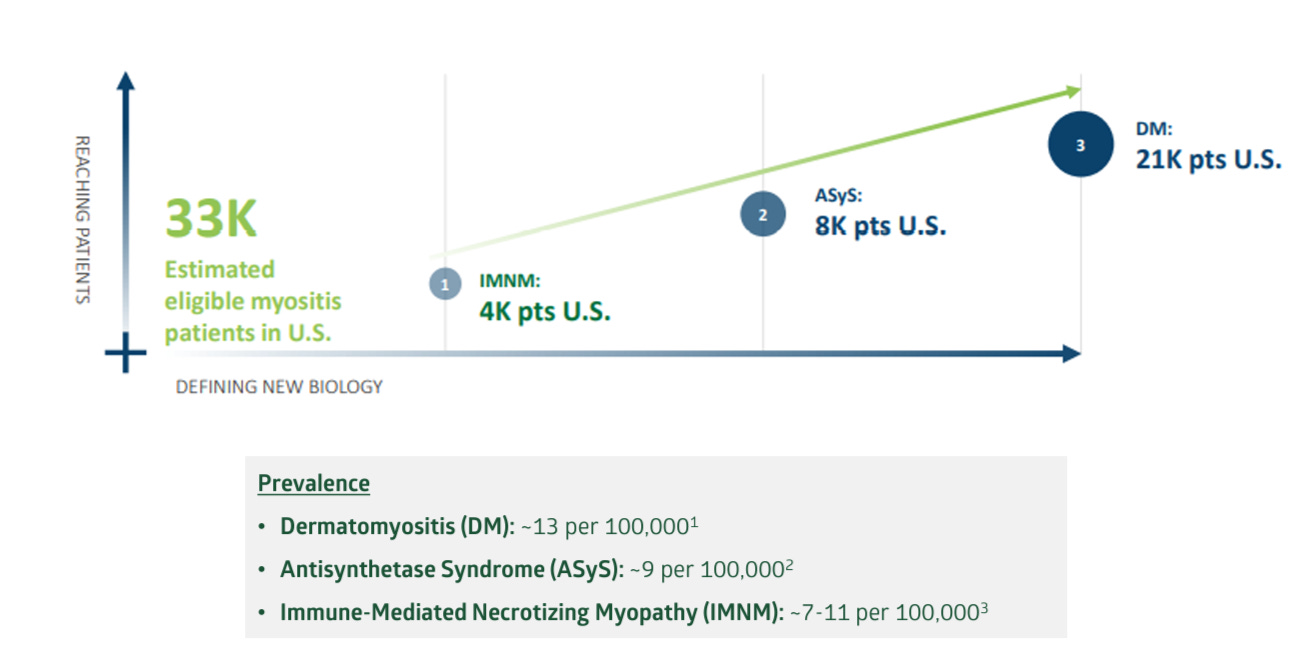
Priovant's strategy involves targeting less crowded therapeutic areas and leveraging Brepo's unique properties. Success in these areas could significantly increase the drug's value, despite the setbacks in lupus treatment. The market potential, especially in dermatomyositis, provides an opportunity for substantial revenue, underlining Priovant's potential as a valuable component of ROIV's portfolio.
Genevant
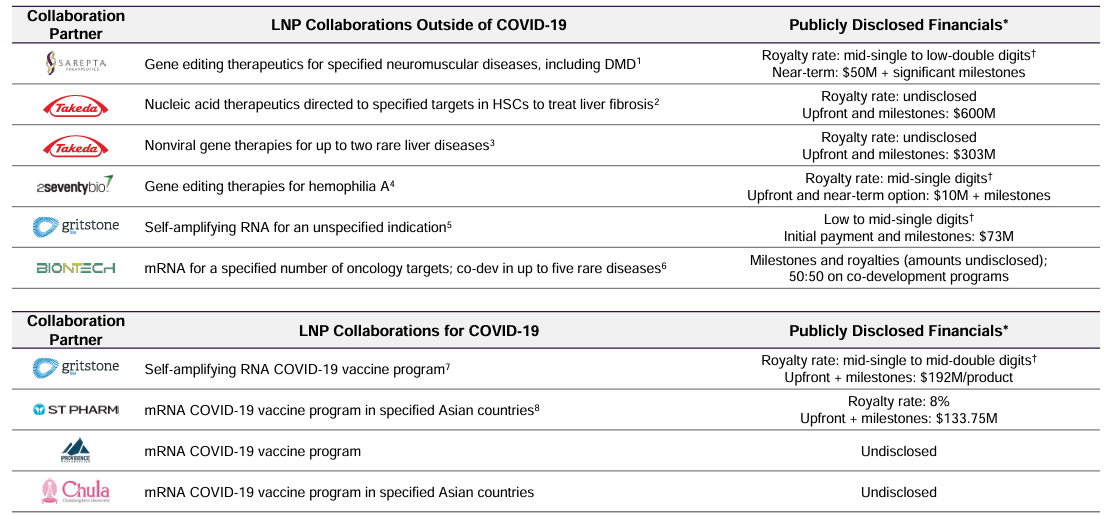
Genevant's legal battles against major pharmaceutical companies over lipid nanoparticle (LNP) technology could result in significant financial compensation. Genevant claims rights to LNPs used in mRNA COVID vaccines, and successful litigation could lead to billions in damages. The complexity of the patent landscape and the potential outcomes of these lawsuits represent both a risk and an opportunity for ROIV.
Dermavant
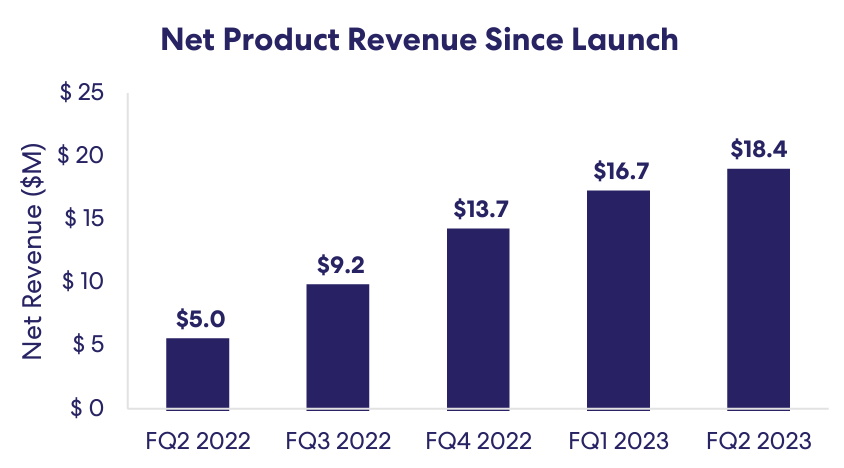
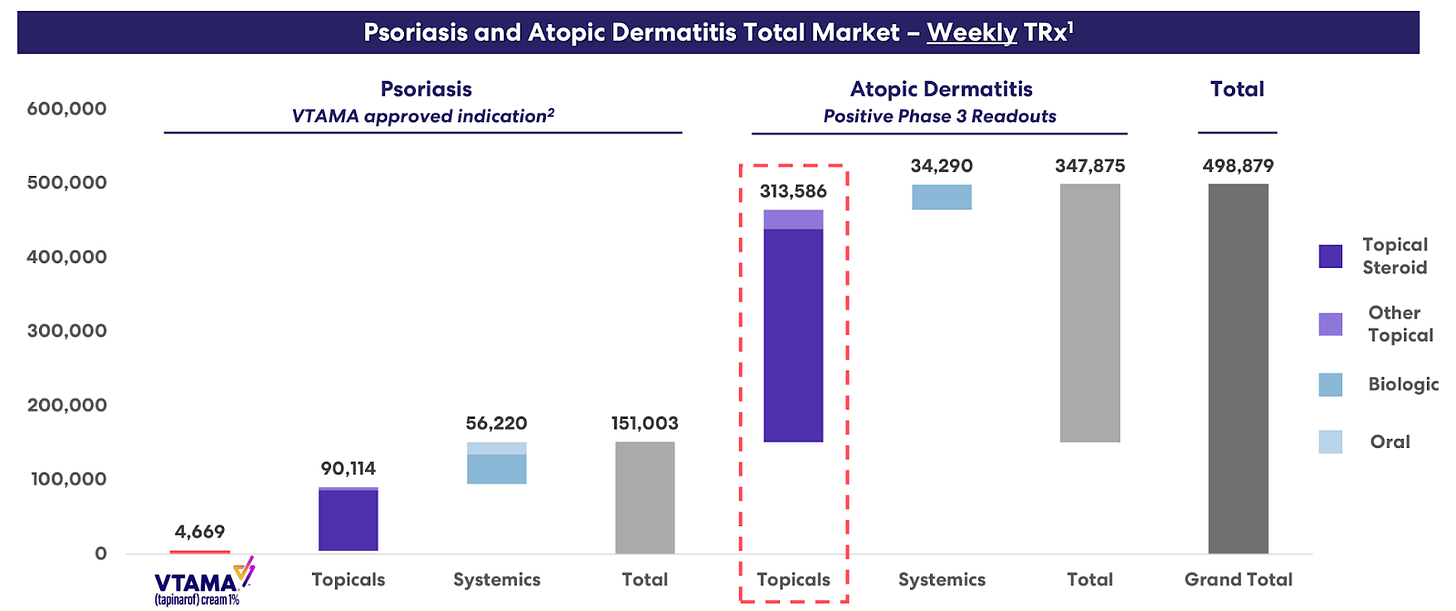
Dermavant, with its product VTAMA, addresses a large market in dermatological conditions. Despite high launch costs, the potential for VTAMA to replace topical steroids due to fewer side effects presents a significant growth opportunity. Achieving a modest market share could lead to substantial revenues, highlighting Dermavant's importance in ROIV's portfolio.
Datavant
Datavant, a health data clearinghouse, plays a crucial role in streamlining patient data for various stakeholders in healthcare. Its recent merger and potential in facilitating drug trials underscore its value. However, the opacity in its operational details and current valuation pose challenges in fully assessing its worth.
Minor Vants'
The Minor Vants, including Kinevant, Hemavant, Covant, Psivant, and VantAI, represent early-stage ventures with diverse focuses ranging from drug development to AI-driven drug discovery. Their current contributions to ROIV’s portfolio may be limited, but they hold significant potential for future growth and value creation. Below is an abbreviated summary for each of these, since they don’t contribute a ton of value right now, nevertheless they still have significant optionality that is completely ignored by the market.
Kinevant - Kinevant is developing Namilumab, a GM-CSF inhibitor for sarcoidosis. Sarcoidosis is an immune driven inflammatory disease suffered by ~150k people in the US, and typically affects the lungs but can affect other organ systems as well. Treatment today consists largely of immunosuppressants (which has negative effects on overall health) and organ specific drugs to alleviate symptoms (such as joints). Granulocyte macrophage colony stimulating factor (GM-CSF) is implicated in the development of sarcoidosis’s granulomas, so the theory is the inhibition of that factor will lead to a reduction in symptoms and tissue damage.
Kinevant dosed the 1st patient in their 100 patient phase 2 in 2022, with results expected in 2H 2024.
Hemavant - Hemavant is developing RVT-2001, an inhibitor of SF3B1. This one gets into some SCIENCE so bear with me. SF3B1 is a spliceosome protein that is mutated in ~80% of MDS-RS (myelodysplastic syndrome with ring sideroblasts). While the mutation is actually associated with positive prognostic value (i.e. lower risk disease, which still has an expected survival of only ~5 years), it causes ineffective erythropoiesis, which results in both low sodium and low iron conditions in patients. Today these conditions are treated by erythropoiesis-stimulating agents (ESAs), but eventually do progress to becoming red blood cell dependent over time (i.e. you require blood transfusions for life).
There is some data for Hemavant, which can be found here (https://ashpublications.org/blood/article/134/Supplement_1/673/426543/Results-of-a-Clinical-Trial-of-H3B-8800-a-Splicing) that shows in an early study 14% of patients treated with RVT-2001 (then called H3B-8800) had decreased requirements for transfusions.
Covant - Ill keep this brief. Covant was incubated at ROIV to accelerate drug discovery through the use of proteomics (basically next-gen mass spectrometry) to optimize proteins for covalency. In March 2023 they announced their first partnership with Boehringer Ingelheim on the discovery of drugs targeting ADAR1, where Covant received $10M upfront, plus $471M in additional milestones and tiered royalties on global sales. Covant is so early we only include a range of values from $0 – $50M today, but it could provide additional optionality over time as the opportunity becomes more clear.
Psivant - Psivant was created through the acquisition of Silicon Therapeutics in 2021, for $450M in ROIV stock plus additional milestone payments. Silicon Therapeutics had built a platform for developing small molecule drug candidates using computational methods and their own supercomputer. ROIV has indicated they would not do another deal of a “platform” like this, and while they are happy they own it and do think it will eventually result in attractive new assets, management acknowledges the price they paid was too high and the market no longer values computational discovery platforms like they once did.
VantAI - VantAI is another asset that could be very exciting given they are in a hot area (AI-driven drug discovery with a focus on molecular design) and have high profile partners (JNJ, Boehinger Ingelheim, Blueprint Medicines). The flip side of this is without clear assets or economics behind these partnerships, it’s nigh impossible to value it with any level of accuracy. So while we have a placeholder valuation today, I believe ROIV is planning to raise venture funding at VantAI in the next 12 months which will hopefully put some numbers around what the asset could be worth.
Lokavant - Lokavant is effectively a software dashboard to accelerate and improve clinical trial enrollment. Conducting an effective trial on time and on budget is critically important to any biotech, so ROIV developed their own tools to optimize how those are monitored (keep in mind, the trials themselves are largely conducted by academic hospitals, but the company works with those trial sites to ensure both speed and trial integrity are progressing according to plan). Technical Dynamics
Technical Dynamics and Conclusion
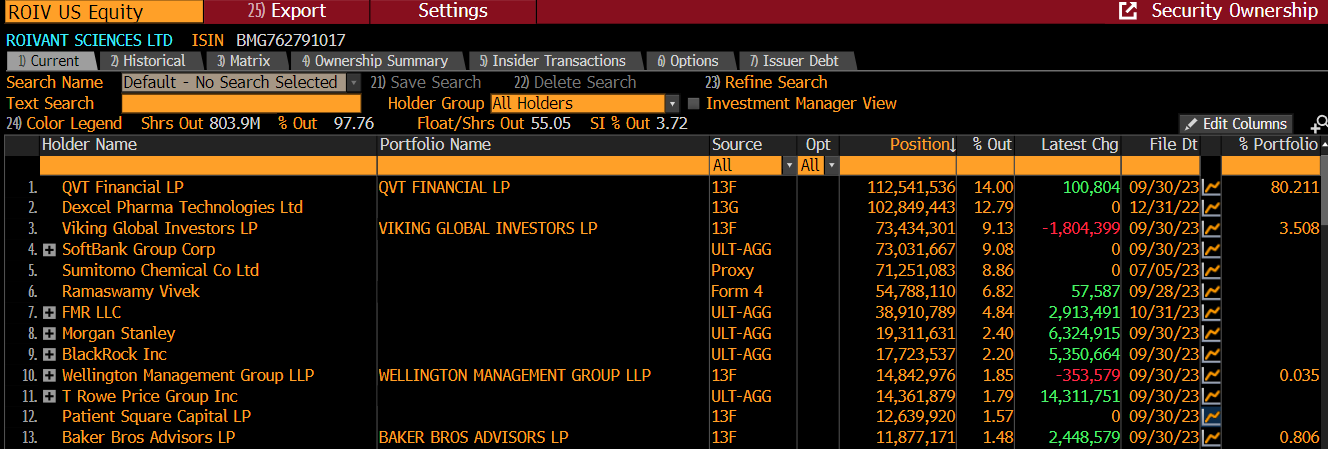
QVT - Vivek’s former employer, which has now converted into a family office. They have board representation and appear only to sell when they need liquidity to manage the wind down of their remaining LP commitments (so far roughly 1x per year).
Dexcel - The largest private pharmaceutical company in Israel, the Co-CEO of which is the Chairman of ROIV’s board of directors. They participated in the seed financing of ROIV and have never sold.
Viking - Invested in 2016 in their private fund. Have sold modestly this year, but based on our understanding they did so for position size reasons and are not sellers at the current valuation.
Softbank - Invested $1.1B in 2017 and have sold periodically, largely in keeping with the woes of the Vision Fund more broadly. Ultimately we think they continue selling periodically, but apparently have not been sellers post the sale of Telavant despite the end of their lock-up in early November.
Sumitomo - The most obviously problematic shareholder today. They have publicly expressed the goal of reducing leverage through the sale of up to ¥150B in cross holdings and equity method investments, the largest liquid of which is ROIV shares. Sumitomo is locked up until February 2024.
Vivek - Personally I am of the view that Vivek only sells if forced, specifically if forced because he has been appointed to a Republican cabinet and therefore gets to avail himself of the same federal loophole (Section 1043) that Hank Paulson took advantage of when appointed Treasury Secretary: “individuals who are forced to sell stock to meet federal conflict-of-interest rules to defer paying capital gains tax, so long as the proceeds are reinvested in government bonds, diversified index funds and other similar instruments”. While I appreciate this theory is slightly conspiratorial, the fact he defer taxes indefinitely might explain, in part, Vivek’s presidential ambitions.
ROIV represents a compelling R/R situation at current levels, with substantial optionality embedded across the portfolio, the cash to see that optionality through to value inflection points, and a management team that is pragmatically focused on asset light development and the eventual return of capital. Perhaps the most underappreciated part of the ROIV story is that the unknown pieces very well might end up being the most valuable.
Catalyst
1. Near term capital return in the form of buybacks from Telavant proceeds
2. Sale of IMVT
3. Sale of WholeCo w/ IMVT
4. Clinical success on existing optionality
5. New partnerships / Vants


to the moon